A topic of increasing interest and importance for dairy cattle production is the effects of endotoxins on performance parameters. It is becoming more evident that endotoxins play a role in the incidence of infectious and metabolic diseases in dairy cattle, impacting health, performance, and profit.
Endotoxins and the gastrointestinal tract
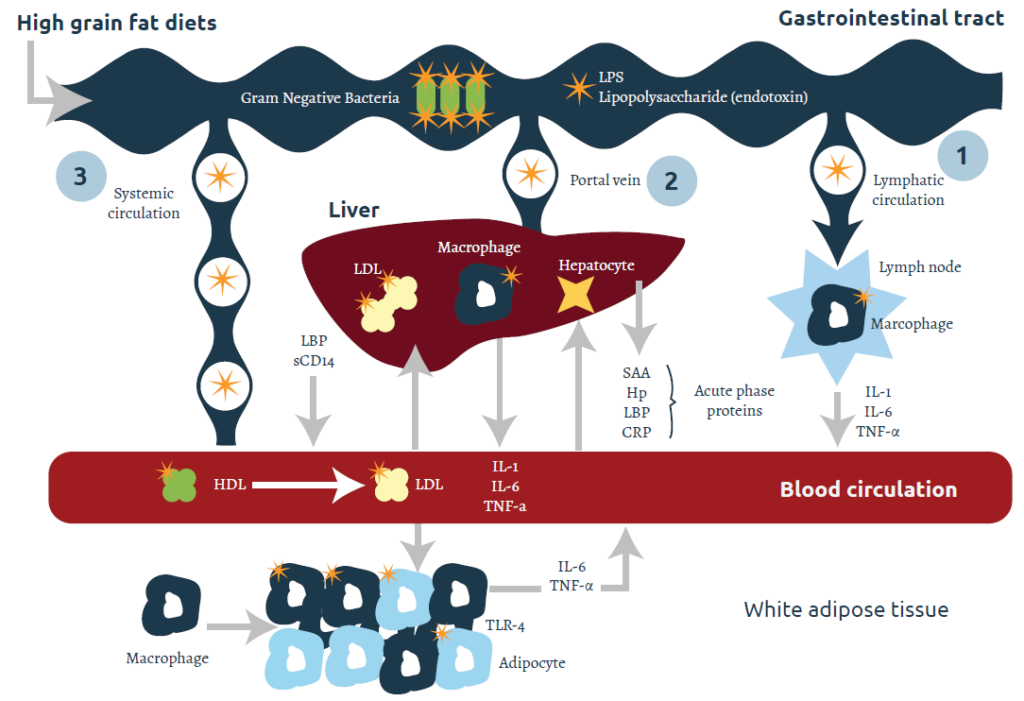
Endotoxins, or commonly called lipopolysaccharides (LPS), are components of the outer cell wall of gram-negative bacteria which are released during bacterial growth. Under normal physiological conditions, the epithelium acts as a barrier to prevent LPS entry into the blood circulation so that only a limited quantity of LPS penetrates the gastrointestinal epithelial barrier (Andersen et al., 1994).
Changes in physiological conditions, such as stressors caused by antigens, microorganisms, cytokines, pH fluctuations, dietary modification, lactation stage, and psychological stress, may cause impairment of the barrier properties of tight junctions, resulting in increased epithelial permeability and the uptake of LPS (Gareau et al., 2008). Differences in the epithelium structure between the rumen and the intestines may translate into different susceptibilities for LPS translocation.
Translocation, or transport of LPS across the epithelium, may occur across the epithelial cell membrane (transcellular) and/or across the tight junction between cells (paracellular) (Tomlinson and Blikslager, 2004). Transcellular transport mainly occurs by active mechanism via receptors on the apical membrane, triggering receptor-mediated endocytosis.
Endotoxins produced in the GIT have three potential entries into the system: the lymphatic system, the portal vein, or systemic circulation (Figure 1). The increase in pro-inflammatory cytokines (PIC) IL-1, IL-6, and TNF-α concentrations in circulation may originate from damage to the gastrointestinal epithelium, macrophages in the liver (Kuppfer cells), lymph nodes, adipose tissue, or a combination. The production of these pyrogen PICs causes metabolic, clinical, and immunologic effects.
Translocation of endotoxins through the gastrointestinal tract
Endotoxins and etiopathology
There is growing knowledge about the role of endotoxins in the etiopathology of multiple periparturient diseases of dairy cows: laminitis, retained placenta, metritis/endometritis, fatty liver, displaced abomasum, and milk fever. However, it should be taken into account that these conditions are multifactorial, meaning that a single factor is not solely responsible for their onset.
The contribution to disease is either direct, through triggering an inflammatory response, or by other factors, such as the overreaction of the natural protective mechanisms of the host (Eckel and Ametaj, 2016). Not only the concentration of endotoxins but also the toxicity of the bacterial species plays a role in the induction of inflammation. For example, the endotoxin of E. coli is much more toxic when compared with other gram-negative bacteria found in the rumen of cattle (Hurley, 1995), meaning that when the LPS of E. coli predominates the rumen, the negative effect is expected to be greater.
Endotoxins and SARA
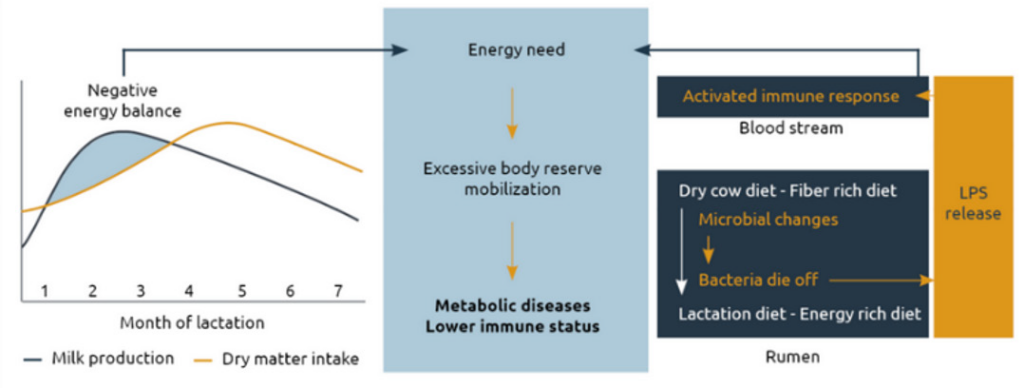
During transition, the system of a dairy cow is under a lot of pressure (Figure 2). Early lactation is characterized by a prolonged negative energy balance and significant changes in dietary intake. To meet the energy demands to produce milk, the ration is changing from a low energy and high fiber diet in the dry period, to high grains and low fiber after calving. However, in many cases, the provision of extra energy is associated with a substantial increase of ruminal and intestinal endotoxin load, which can reach systemic circulation and have consequences on production.
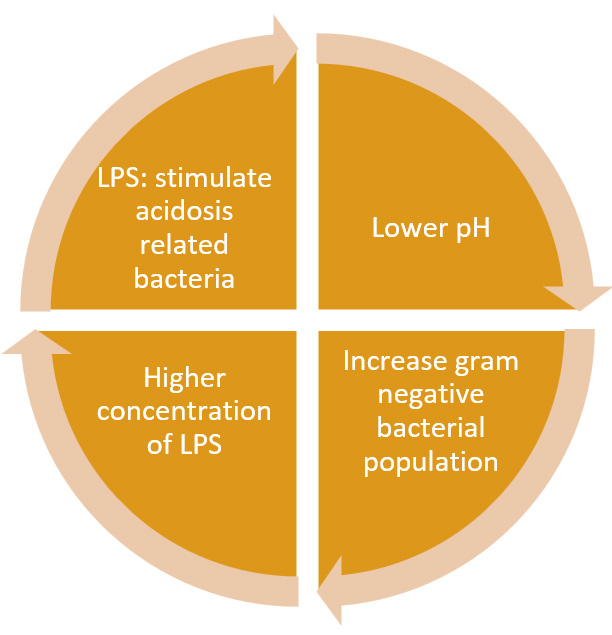
One of the most common feeding disorders in dairy cows is subacute rumen acidosis (SARA), with an overall prevalence between 11–26% (Plaizier et al., 2018). The term SARA is often used as a synonym for poor rumen health and it is defined as a threshold of 180 min/day below pH 5.6. A low pH in the rumen causes a decrease in bacteria richness (Plaizier et al., 2017), changes in bacteria attached to rumen epithelium (Petri et al., 2020; Plaizier et al., 2021), and an increase in gram-negative bacteria populations. LPS are released as a result of the increased bacterial growth rate or due to the acidity which makes bacteria more prone to lysis/rupture. Therefore, a higher concentration of free LPS is detected in the rumen fluid of cows experiencing SARA (Emmanuel et al., 2007; Zebeli et al., 2011; Zhou et al., 2014). LPS may contribute to SARA since LPS can stimulate the growth of acidosis-related bacteria like Streptococcus bovis and Selenomonas ruminantium (Dai et al., 2020) to create a vicious circle (Figure 3).
Endotoxins and a multifactorial preventative approach
As explained in the previous sections, due to the close relationship between SARA and endotoxicosis, practices to prevent SARA and improve rumen health will help. The importance of adequate dietary fiber, type of concentrate and starch, and feeding management practices are described in the literature. However, in intensive dairy farming, SARA is difficult to avoid so, it is important to further prevent endotoxin’s negative effects and keep dairy cows healthy with a broad spectrum of support.
The impact of endotoxins in productive parameters is the result of their concentration and toxicity, the integrity of the epithelium, changes in rumen fermentation profiles, translocation of endotoxins, immune response, changes in nutrients partitioning, and shifts in behavior. First of all, with the use of the toxin binder, Vitafix® Spectrum, the concentration of endotoxins and mycotoxins in the GIT can be reduced. his dual benefit is a cost-effective method to decrease the chance of toxins interacting with the pithelium, preventing them from entering the systemic circulation and causing an inflammatory response. In addition, the gastrointestinal epithelial wall forms a barrier that can be compromised due to different factors, like LPS. With Vitanox, this can be prevented by having a positive effect on the integrity of epithelial cells in the small intestine. Finally, stabilizing the rumen microflora with medium-chain fatty acids (Aromabiotic® Cattle) helps to create a healthy rumen environment. This leads to fewer endotoxins being released, reducing the activation of the immune system, and sparing energy to improve milk production.
Strengthening the cow’s general health and immune status allows for multiple healthy lactations. Increasing longevity with preventative measures is a sustainable solution to maximize the profitability of dairy cows and the income for the farmer.
References are available upon request.